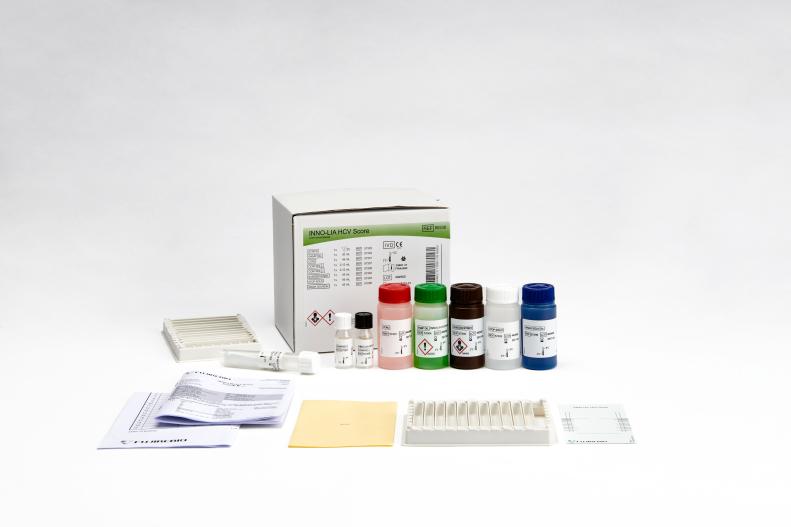
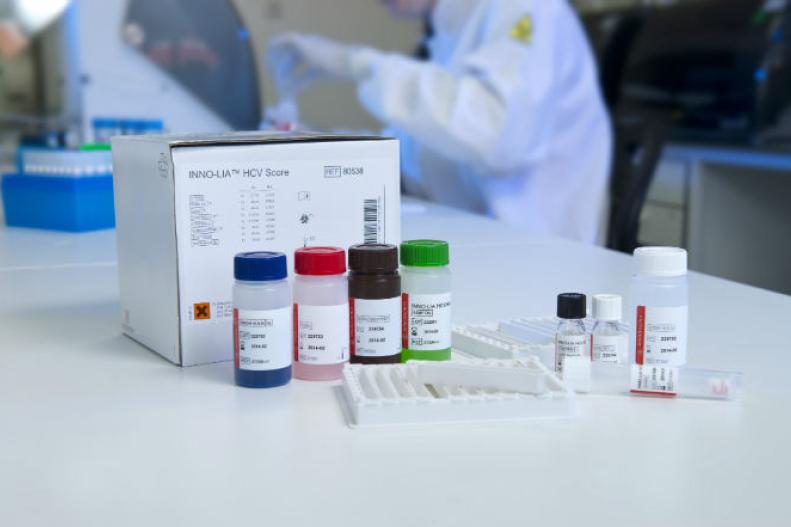
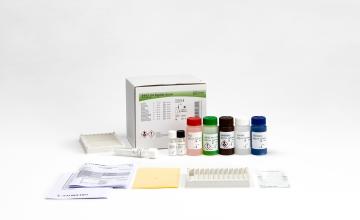
INNO-LIA® HCV Score
Optimal coverage of immunodominant HCV epitopes using unique and specific antigens
Line immunoassay for the detection of antibodies to human hepatitis C virus in human serum and plasma. It is intended for use as a supplementary test on human serum or plasma specimens found to be reactive in an anti-HCV screening procedure.
The INNO-LIA HCV Score assay utilizes well-defined antigens derived from HCV immunodominant proteins from the core region, the E2 hypervariable region (HVR), the NS3 helicase region and the NS4A, NS4B and NS5A regions. The antigens used are either recombinant proteins or synthetic peptides, highly purified, and fixed on a nylon membrane.
INNO-LIA® HCV Score
Product number 80538
20 Tests
CE marked (IVDR)
Please contact your local Fujirebio representative for the availability of this product in your country.